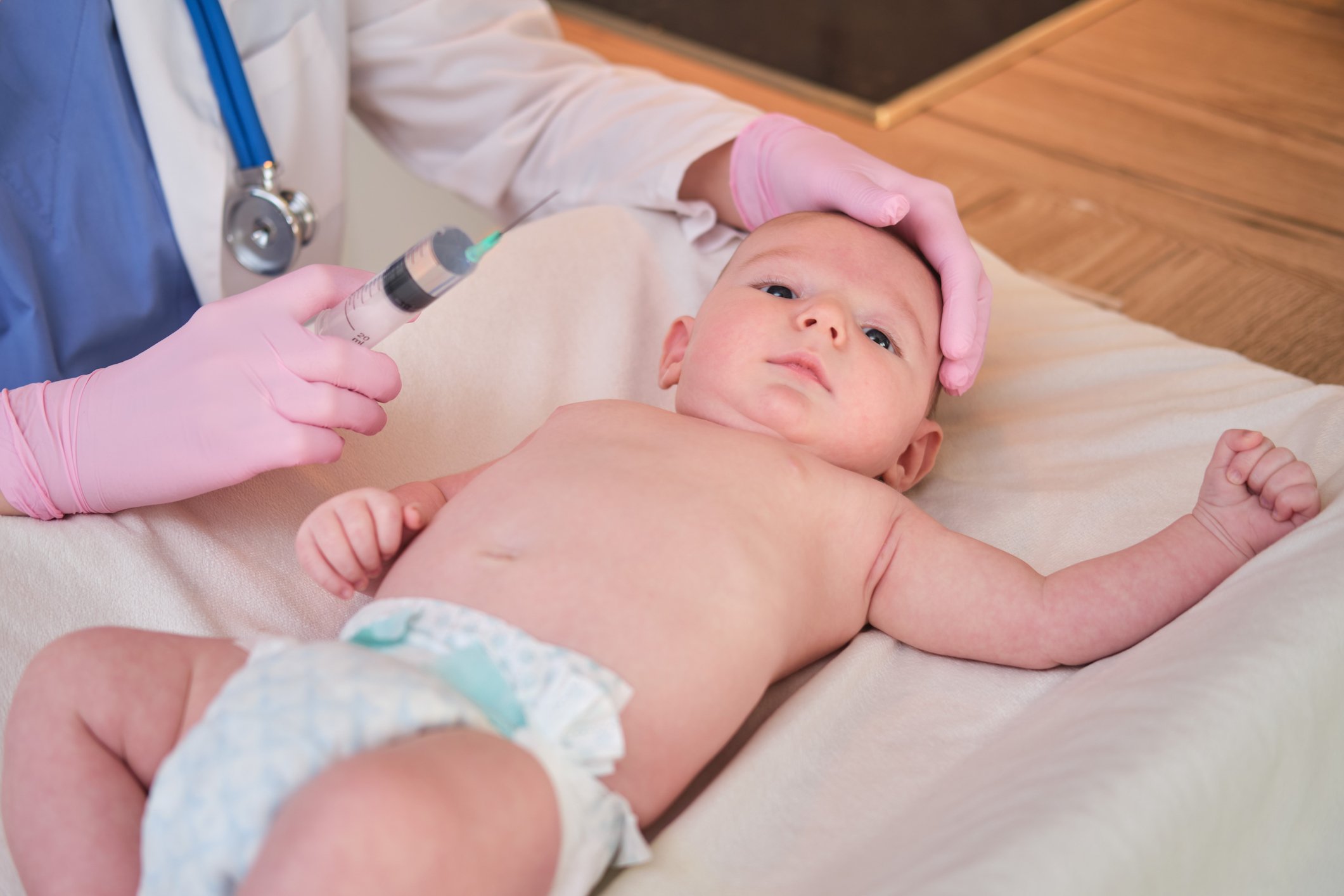
New data is in on the fallout from a controversial vaccine policy change, and the numbers are stark. Two studies published Monday in JAMA Pediatrics project that delaying the hepatitis B birth-dose vaccine will lead to hundreds of additional infant infections, chronic cases, and deaths — with the true toll likely higher once real-world adherence gaps are factored in.
The policy, adopted by the Advisory Committee on Immunization Practices at the end of 2025, replaced the universal recommendation for a hepatitis B vaccine at birth with a “shared clinical decision-making” approach for infants of hepatitis B-negative mothers, effectively allowing the first dose to be pushed to two months of age. The committee voted after being swept clean by the Health and Human Services secretary and stocked with appointees who have histories of vaccine skepticism.
One of the new studies modeled a single U.S. birth cohort of 3.6 million infants. Even with perfect adherence to the new guidelines, a two-month delay is projected to cause 90 additional acute hepatitis B infections, 76 chronic infections, 29 deaths from hepatitis B-related causes, and $16.4 million in added lifetime healthcare costs. The numbers escalate when accounting for infants whose mothers' hepatitis B status is unknown or when adherence falters — as it inevitably does.
The second study looked at what happens when universal vaccination disappears. History offers a grim precedent: In 1999, a similar delay recommendation caused birth-dose coverage among infants of unscreened mothers to collapse from 53 percent to 7 percent before the policy was reversed. Applying that floor to the current change yields 628 additional infant infections per cohort. About 90 percent of perinatally acquired hepatitis B becomes chronic, and one in four of those children will die prematurely from cirrhosis or liver cancer.
Cascade of Consequences Beyond Hepatitis
The direct infection numbers are only part of the story. The same “shared clinical decision-making” framework that weakens the hepatitis B vaccine also threatens two other universal newborn interventions: intramuscular vitamin K and erythromycin ophthalmic ointment. Pediatric literature consistently shows that when one intervention is refused, parents often refuse all three.
Vitamin K deficiency bleeding, which typically presents at four to eight weeks of life as catastrophic intracranial hemorrhage, carries a mortality rate near 20 percent. Survivors often suffer severe neurological damage. Clusters of this condition have already been reported in the U.S. and Australia in areas with active vaccine skepticism. Ophthalmia neonatorum, untreated, blinds a newborn within days. Erythromycin costs pennies, but refusal rates are climbing alongside vaccine refusals.
The pattern echoes earlier public health missteps. In April 2020, a sitting president mused about injecting disinfectants, triggering a spike in poison-control calls. After an offhand remark about hydroxychloroquine, an Arizona man died from drinking a fish-tank product containing chloroquine. More recently, the HHS secretary recommended vitamin A and cod liver oil during the West Texas measles outbreak. Two unvaccinated children died — the first U.S. measles fatalities in a decade. At Covenant Children’s Hospital in Lubbock, doctors documented hypervitaminosis A in measles patients with elevated liver enzymes, after parents dosed their children at home with the secretary’s recommended remedy.
Congress Built the Firewall; Now It Must Defend It
The Advisory Committee on Immunization Practices is not merely advisory in the colloquial sense. By statute, its recommendations determine eligibility for the Vaccines for Children program and trigger no-cost-sharing coverage under the Affordable Care Act. When the committee weakens a recommendation, federal vaccine financing and private insurance coverage weaken with it. The committee was created by Congress, and its composition and independence are now under scrutiny.
The Senate HELP Committee and House Energy and Commerce Committee have oversight authority but have used it sparingly. Hearings on the committee’s membership, conflict-of-interest screening, and the evidentiary basis for the December vote are overdue. Legislation to insulate committee members from secretarial dismissal without cause is also needed. Inspector general protections were supposed to do that work but failed.
As one UCLA intensive care unit physician with over 40 years of experience put it: “Science survived bad presidents and bad administrators. But it will not survive a captured advisory committee without a fight from the branch that built it.”
Tonight, a parent will look down at her newborn and say, “Let’s wait.” But the virus will not wait, and neither will the bleed.